
As a maker of medical devices, there are a number of regulatory bodies that you must work with to successfully bring your product to market.
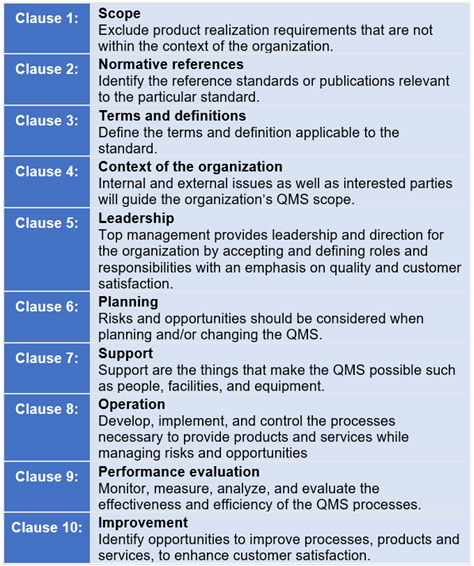
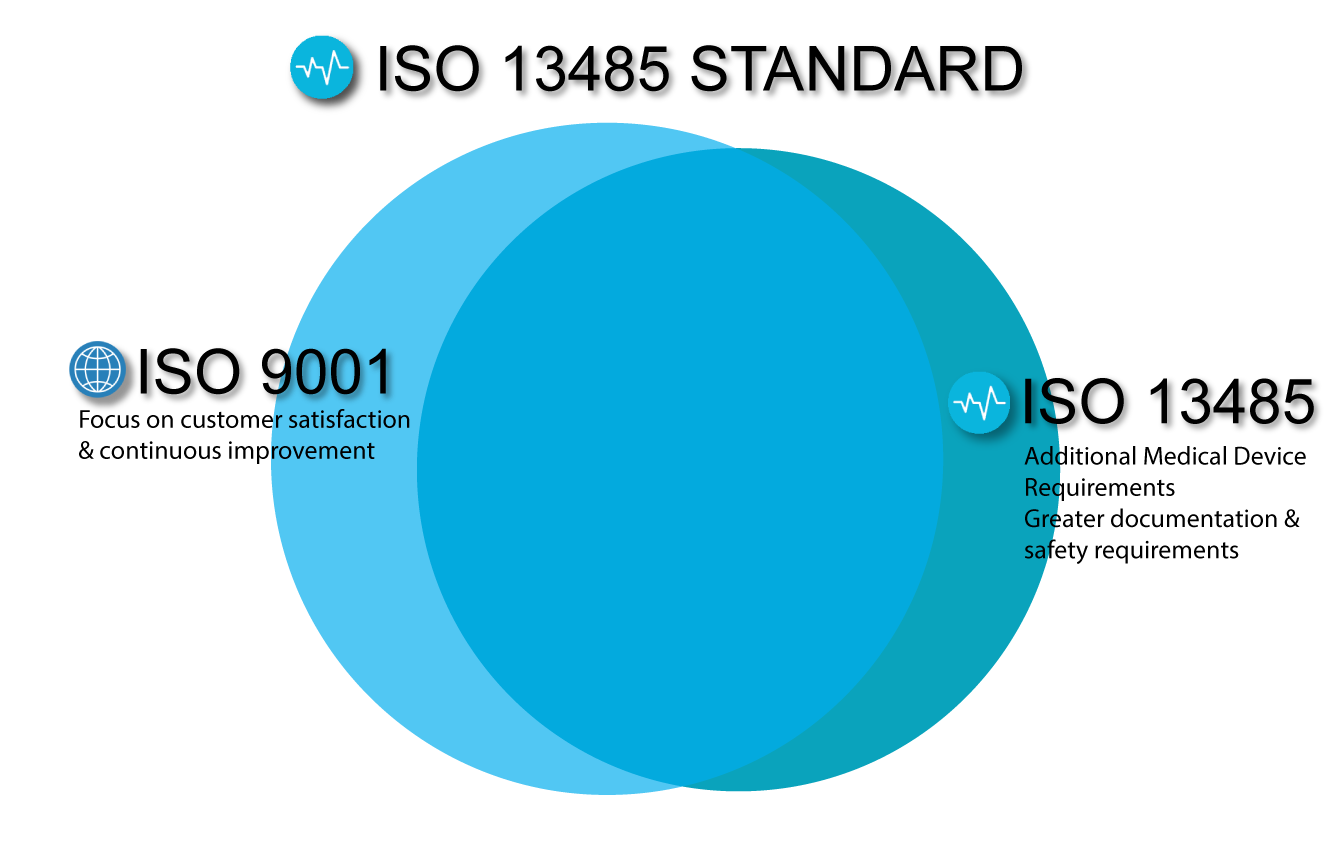
When an organization implements a QMS, it means it has documented processes, procedures, and responsibilities in place to achieve quality objectives and policies. It outlines specific requirements for implementing a QMS at an organization involved in the design, production, installation, and servicing of medical devices, as well as any related services. ISO 13485 is the quality management system (QMS) standard for the medical devices industry. Here’s a closer look at the ISO 13485 certification key elements and how having that certification in place by your PCBA contract manufacturer will benefit you. That includes your contract manufacturer for your PCBAs. Not only must your organization be certified to this quality management standard for the medical devices industry, but it is important to ensure your entire supply chain is also following these stringent requirements. One of the most important standards, however, is ISO 13485. When you’re in the medical device industry, certifications are critical, as there are several standards and regulations you must adhere to when marketing products in this space. Certifications provide consumers with the assurance that the product or services that they are using will perform or give them the results that they expect. Whether you are in an elevator or at the doctor’s office, one of the first things that you will probably notice is a certification of some sort hanging on the wall.

